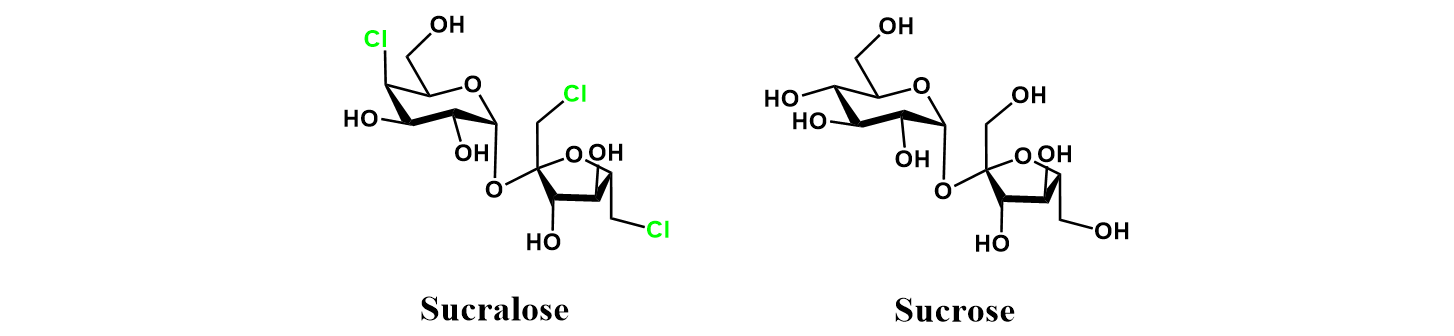
Sucralose is a widely used artificial sweetener. It is noncaloric, and one of the sweetest sugar substitutes currently available, being around 600 times sweeter than sucrose [1]. Commercial sucralose preparations are sold under various brand names, such as Splenda. Sucralose is manufactured from table sugar (sucrose) through selective chlorination. In this article, I explore several patented routes for the industrial production of sucralose from sucrose. In doing so, I illustrate how there are often multiple strategies through which a synthetic organic chemistry problem can be solved.
The chemical structure of sucralose is highly similar to sucrose. Relative to sucrose, it is chlorinated at the C4 position of the glucose unit, and the C1/C6 positions of the fructose unit. Achieving this pattern of selective chlorinations is synthetically difficult. The biggest challenge is on the glucose moiety: chlorinating at C4 without modification at C6. In general, primary alcohols are much better nucleophiles than secondary alcohols, owing to their low steric bulk. Both desired chlorinations on the fructose moiety are primary alcohols. However, on the glucose we require the chlorination of a secondary alcohol (C4) without modification of a primary alcohol (C6).
Control of chlorination by protecting groups [2]
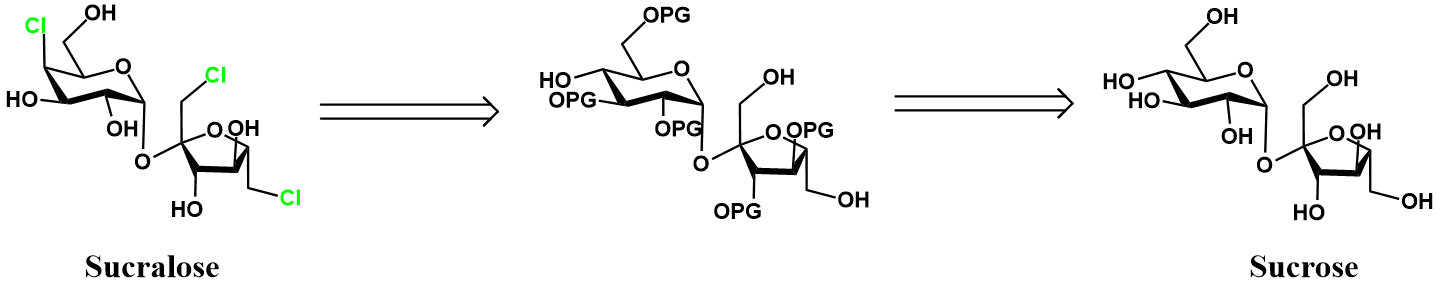
The principle challenge with sucralose synthesis is selectivity. Sucrose has many hydroxyl groups, but we only want three of them to react with chlorinating agent. The most intuitive strategy to achieve this selective chlorination at the required positions is to protect all other hydroxyl groups on sucrose with protecting groups. Retrosynthetically, this approach is depicted below.
The key protected intermediate has protecting groups (PG) at all positions save C4 on glucose and C1/C6 on fructose. The chosen protecting group must be stable to the chlorination conditions and feasible to remove without loss of the chlorine substituents. This protected intermediate can readily be chlorinated and deprotected to give sucralose. The protected intermediate is prepared from sucrose by a series of protecting group manipulations. A complete synthetic scheme for these transformations is given below.
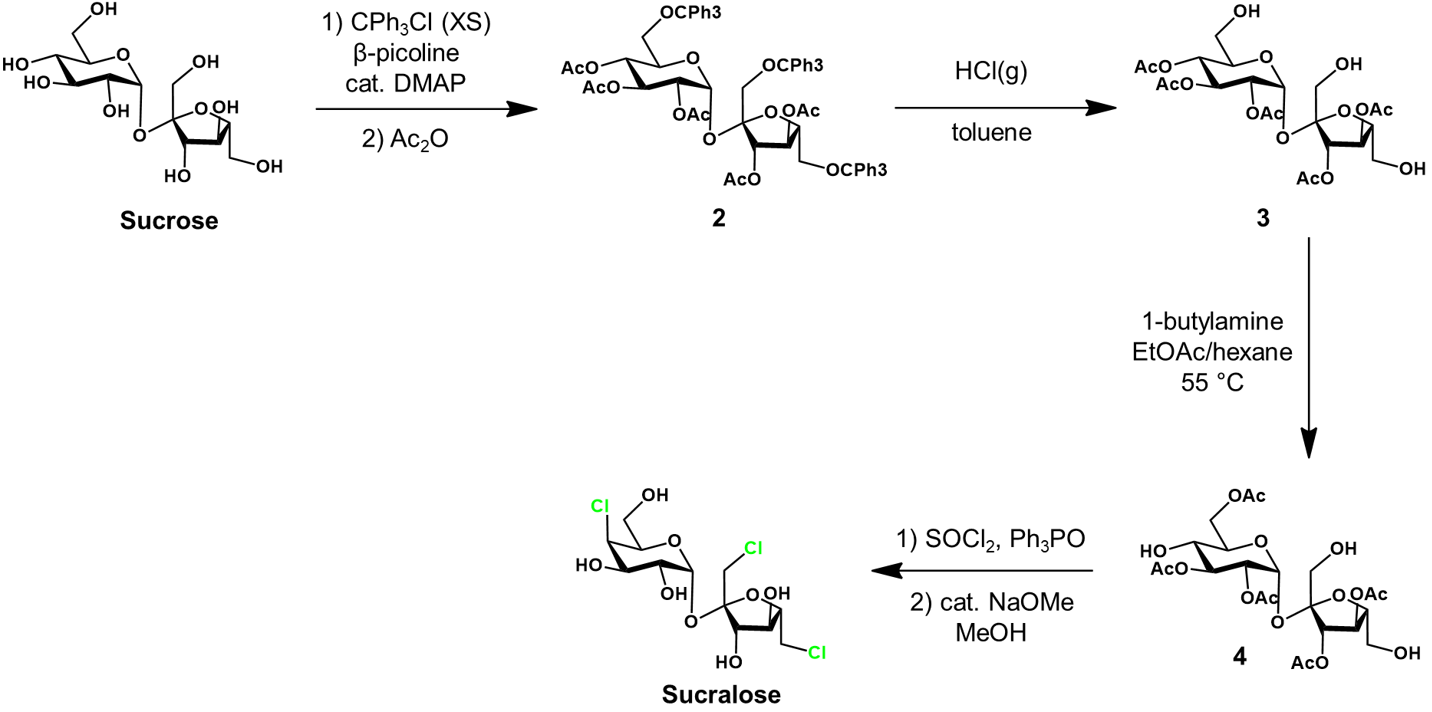
Acetyl groups are a convenient protecting group, since they are acceptably stable to the acidic chlorination conditions and can be efficiently and cleanly removed. However, installing the acetyl groups selectively in the required positions (4) takes a series of manipulations - simple peracetylation of sucrose would indiscriminately protect all hydroxyl groups. The synthesis begins by isolating the primary hydroxyl groups of sucrose, through reaction with trityl chloride (CPh3Cl). The large steric bulk of this group limits the reaction to only the primary alcohols. The sugar is then peracetylated with acetic anhydride (Ac2O) to protect the remaining hydroxyl groups, affording 2. The β-picoline serves two roles in these reactions: as a weak base to neutralize the acid generated, and as a solvent. 4-Dimethylaminopyridine (DMAP) accelerates both reactions through action as a nucleophilic catalyst.
Next, the trityl groups are selectively removed by treatment with HCl in toluene, yielding 3. To attain the desired protecting group arrangement, the next step is critical. We require protection at glucose C6 but not C4. This is achieved by allowing acetyl migration from C4 to C6, giving protected intermediate 4. This transformation occurs with high selectively under treatment with 1-butylamine in a mixture of ethyl acetate and hexane.
Protected intermediate 4 is chlorinated with thionyl chloride (SOCl2) and triphenylphosphine oxide (Ph3PO). The thionyl chloride first chlorinates the triphenylphosphine oxide, generating the active chlorinating agent. This condition is gentler and reduces side reactions/product decomposition compared to using thionyl chloride alone. Lastly, the acetyl groups are deprotected under basic conditions in methanol to afford sucralose. Sucralose is conveniently purified by recrystallization from methanol/water.
Next, the trityl groups are selectively removed by treatment with HCl in toluene, yielding 3. To attain the desired protecting group arrangement, the next step is critical. We require protection at glucose C6 but not C4. This is achieved by allowing acetyl migration from C4 to C6, giving protected intermediate 4. This transformation occurs with high selectively under treatment with 1-butylamine in a mixture of ethyl acetate and hexane.
Protected intermediate 4 is chlorinated with thionyl chloride (SOCl2) and triphenylphosphine oxide (Ph3PO). The thionyl chloride first chlorinates the triphenylphosphine oxide, generating the active chlorinating agent. This condition is gentler and reduces side reactions/product decomposition compared to using thionyl chloride alone. Lastly, the acetyl groups are deprotected under basic conditions in methanol to afford sucralose. Sucralose is conveniently purified by recrystallization from methanol/water.
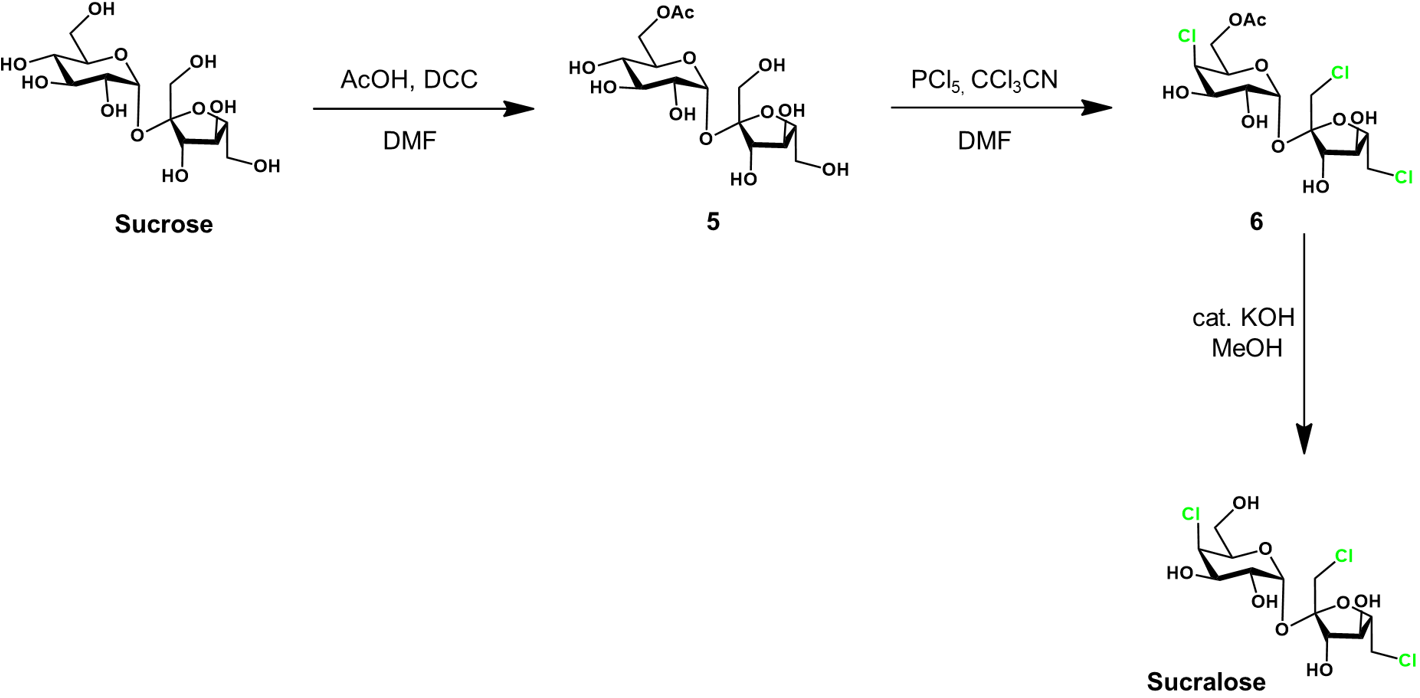
Selective chlorination of sucrose-6-acetate [3]
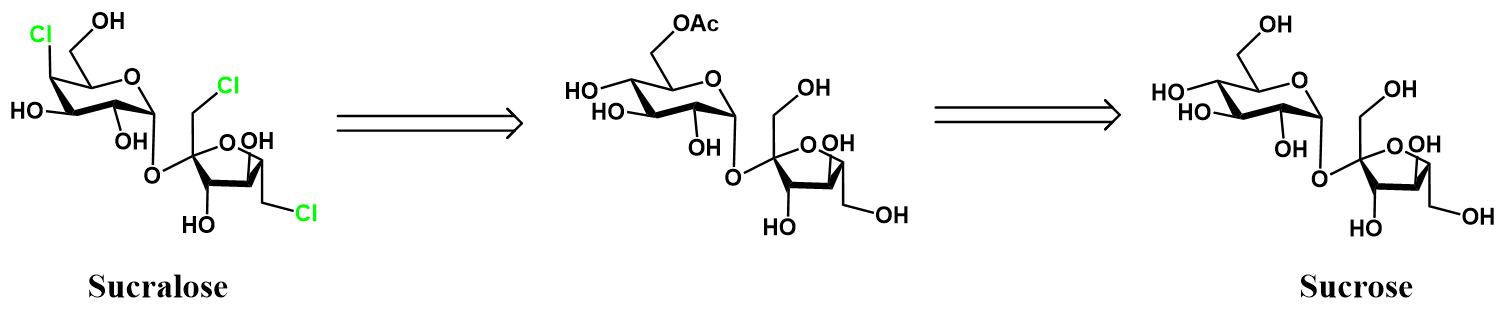
The previous approach to sucralose synthesis was intuitive, but encumbered by a series of protecting group manipulations. The synthesis would be simpler and more efficient if fewer steps were required. This could be achieved if the chlorination was conducted with some selectivity, reducing the protection required. It turns out that such selectivity is possible, requiring only protection of the C6 hydroxyl. This is represented retrosynthetically below.
Sucralose is attained by selective chlorination of sucrose-6-acetate, followed by deprotection of the acetyl protecting group. The sucrose-6-acetate intermediate is produced by selective acetylation of sucrose. A detailed synthetic scheme follows.
First, sucrose is selectively protected at the glucose C6 by an acetyl group, to sucrose-6-acetate (5). This is achieved through a Steglich esterification. The acetic acid (AcOH) reacts with dicyclohexylcarbodiimide (DCC) to form a reactive O-acylisourea intermediate. This activated electrophile is susceptible to nucleophilic attack by alcohols, and reacts with the C6 hydroxyl group with good selectivity. The solvent used for this transformation is dimethylformamide (DMF). As a polar aprotic solvent, it is it useful for promoting SN2 reactions.
The selective chlorination of 5 to 6 is achieved using phosphorous pentachloride (PCl5) as the chlorinating agent. Trichloroacetontrile (CCl3CN) is used as an additive in the reaction, dramatically increasing the reaction rate through an unclarified mechanism. The selectivity attained in this reaction is impressive, likely requiring the carefully optimized conditions. Lastly, the sucralose-6-acetate 6 is deprotected in basic methanol to yield sucralose.
The selective chlorination of 5 to 6 is achieved using phosphorous pentachloride (PCl5) as the chlorinating agent. Trichloroacetontrile (CCl3CN) is used as an additive in the reaction, dramatically increasing the reaction rate through an unclarified mechanism. The selectivity attained in this reaction is impressive, likely requiring the carefully optimized conditions. Lastly, the sucralose-6-acetate 6 is deprotected in basic methanol to yield sucralose.
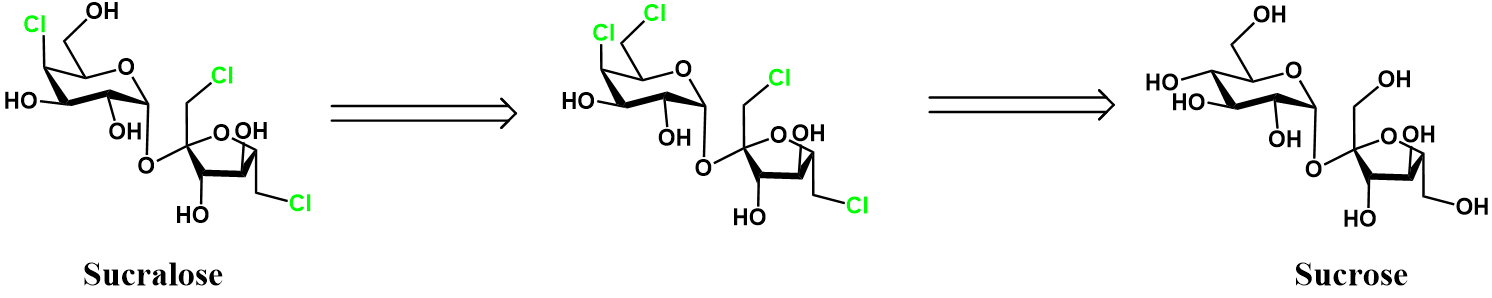
Selective dechlorination of perchlorinated sucrose [4]
Thus far, both approaches to prepare sucralose from sucrose have focused on preventing chlorination of the C6 hydroxyl. A natural alternative to these routes is to over-chlorinate sucrose, then simply remove the unwanted chloride(s). This is depicted retrosynthetically below.
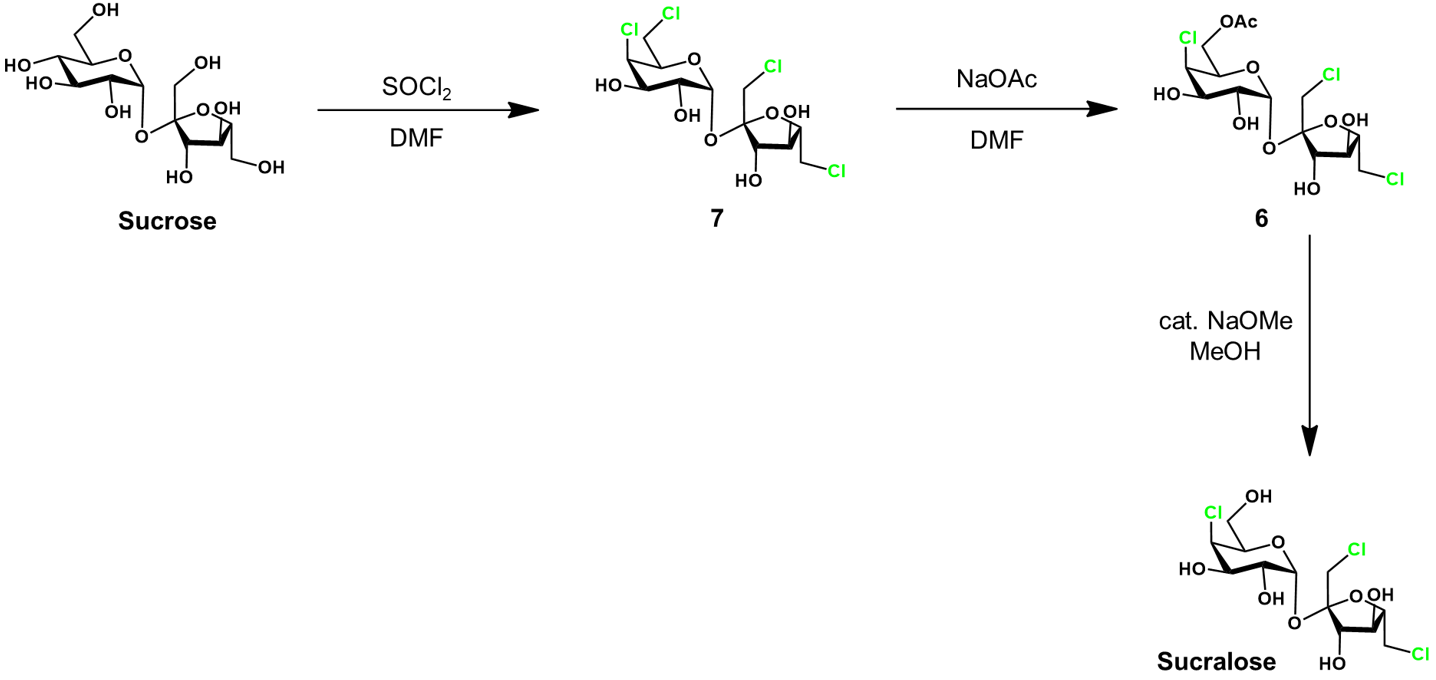
Fortunately, selective chlorination of unprotected sucrose is possible. This spares most of the unwanted chlorination, except at the glucose C6 position. From this chlorinated intermediate, we have to selectively substitute this chloride back to a hydroxyl group. A full synthetic scheme of this route follows.
Sucrose is selectively chlorinated by thionyl chloride in DMF, giving 7. Next, the C6 chloride is substituted to an acetate group selectively by reaction with sodium acetate (NaOAc), yielding 6. It is counter-intuitive that this manipulation would be highly selective for the C6 position of the glucose moiety over the two primary chlorides of the fructose. Nevertheless, this is the specificity that is observed, and it is certainly convenient in this synthesis. As in the previous synthesis, sucralose-6-acetate 6 is deprotected in basic methanol to give sucralose.
 RSS Feed
RSS Feed